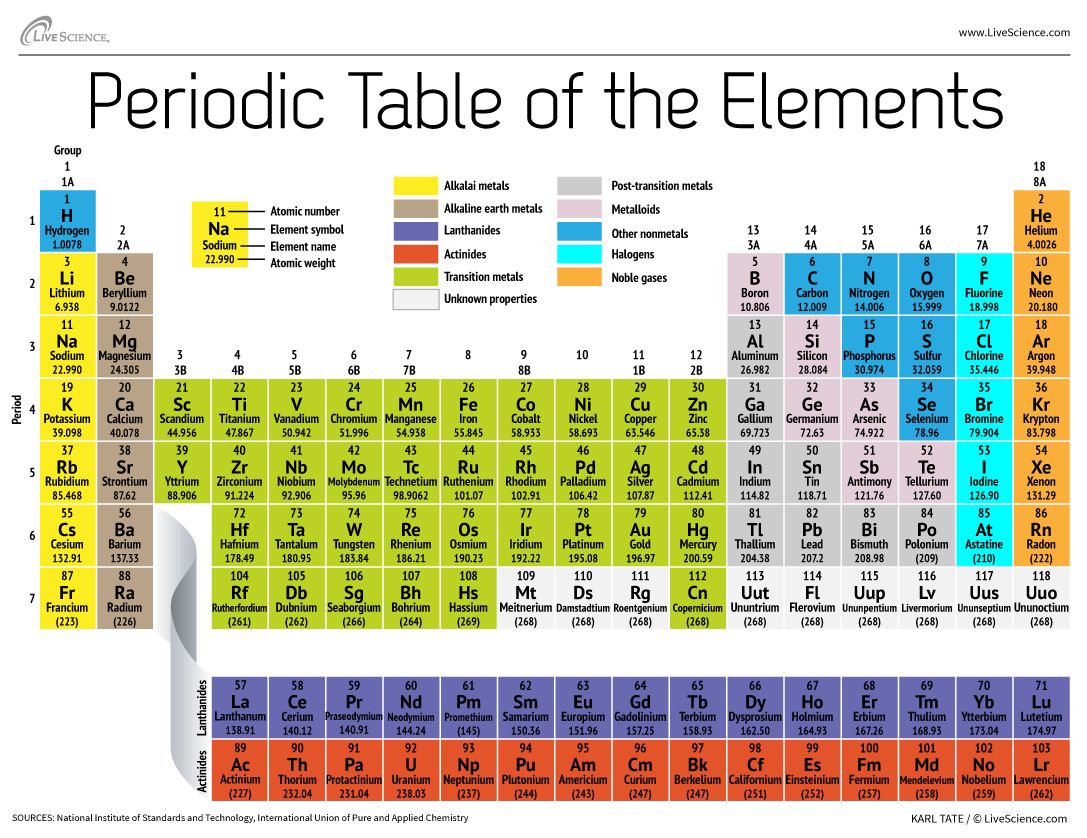
They are found in very minute quantities naturally. These rare earth elements are divided into lanthanides (elements 58 – 71) and actinides (elements 90 – 103). The inner transition or f-block elements are placed under the transition ones in a separate table. The groups 3A, 4A, 5A, 6A and 7A consist of p-block elements. They have similar chemical properties like production of colored compounds and variable valency. The groups are present in the following order: 3B, 4B, 5B, 6B, 7B, 8B, 1B, and 2B contain transition metals or the d-block elements. They contain the same number of outer electrons, and thus show similar chemical properties. Both 1A and 2A contain the s-block elements. The alkali earth metals are found in group one, whereas the second group contains alkaline earth metals. The vertical rows are known as groups and the horizontal rows are known as periods. Thus, the elements are arranged in the order of increasing atomic number (Z) left to right across the table. With this knowledge, he was able to predict the presence of new elements. With these breakthroughs, Mendeleev could not move further as the Rutherford-Bohr model of the atom was yet to be discovered.Īfter about four decades, Henry Moseley in 1913 showed the atomic number (charge) and not the atomic weight, as proposed by Mendeleev, as the fundamental chemical property of any element. It was Mendeleev, who predicted undiscovered elements like eka-silicon (as he found a gap between silicon and tin), today known as germanium, gallium, today called aluminum, and eka-boron, known as scandium. He got a table with gaps and spaces, which meant that there were further elements yet to be discovered. The ones with similar properties were kept in the same column.He positioned element with higher atomic weights on the left side.He arranged the elements in his table on the basis of the following points: Mendeleev found out that 65 elements that were known in his time, could be arranged in a grid. There were many versions introduced before Mendeleev’s table, but he was the one who illustrated the recurring periodic trends in the elemental properties. They follow a pattern, i.e., in an increasing order of the atomic number.ĭmitri Mendeleev, a Russian chemist, is credited as the first person to invent the periodic table in 1869. These elements are arranged in rows and columns – left to right and top to bottom. The elements arranged in the periodic table are great help to scientists, chemists, scholars, researchers, and even students in understanding their various properties and characteristics at a glance. We cover the history of the periodic table thoroughly in our blog post.The periodic table is one of the most important points of reference in the branch of chemistry, and is often known as the Bible of chemical sciences. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. The Periodic Table of Elements can be used as assistance in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. The Periodic table of elements is a tool, developed by scientists over hundreds of years. List of all the elements and the elemental properties:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
